Medi-Globe Launches mAI Companion® — the World’s First Real Time AI Assistant for Pancreatic EUS, Co Developed with IHU Strasbourg —Now MDR CE Marked and Available for Clinical Use in Europe
24.2.2026 10:00:00 CET | news aktuell GmbH | Press Release
Rohrdorf (Achenmühle), Germany — February 24, 2026 — Medi-Globe today announced the launch of mAI Companion®, a real-time medical AI solution designed to assist physicians in detecting pancreatic lesions during endoscopic ultrasound (EUS) — addressing one of the deadliest and most difficult-to-detect cancers, where earlier identification can dramatically change patient outcomes.
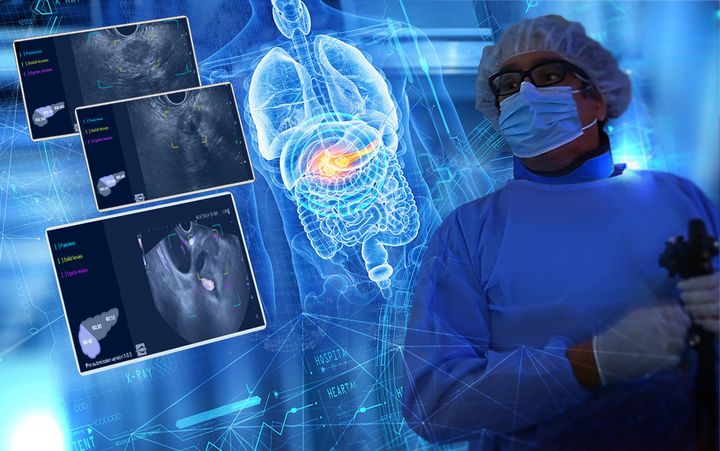
Developed through a multi‑year collaboration with IHU Strasbourg, mAI Companion® has received MDR CE Mark certification, becoming the first‑of‑its‑kind AI assistant for pancreatic EUS approved for clinical use in Europe.
Addressing One of Medicine’s Toughest Imaging Challenges
Pancreatic cancer remains one of the deadliest malignancies worldwide, and early detection can significantly improve survival. EUS provides high‑resolution, close‑up imaging and allows targeted biopsy confirmation—but the technique demands years of experience, and subtle lesions can still be difficult to detect.
mAI Companion® functions as an intelligent “second set of eyes” during EUS, automatically analysing the head, body, and tail of the pancreas in real time to highlight solid and cystic lesions that may otherwise go unnoticed. The expected result: greater diagnostic confidence, more systematic exams, and fewer missed findings supporting earlier identification of pancreatic lesions including stages when treatment options and survival outcomes are significantly improved.
Built on Expert Knowledge — at Unprecedented Scale
AI performance depends on data quality. mAI Companion® was created in collaboration with leading European expert centres and trained on hundreds of real‑patient EUS videos containing more than five million expert‑annotated images. Each annotation was validated by experienced endoscopists, enabling the software to act like a trusted expert advisor beside the clinician.
First‑in‑Human Use, Clinical Evidence
mAI Companion® underwent first‑in‑human clinical use ahead of market introduction—a milestone Medi-Globe first reported in 2022. Patents covering key elements of the technology have been filed.
A tandem randomised EUS video trial involving 57 endoscopists—currently under peer‑review—demonstrated significantly higher diagnostic accuracy and a marked reduction in missed‑lesion rates when using AI compared with standard assessment.
Clear Clinical and Institutional Benefits
The MDR CE‑Marked mAI Companion® is expected to deliver practical value across all levels of clinical practice — improving confidence, consistency, and clinical quality in pancreatic EUS:
- Experienced Endoscopists: Greater reassurance in complex or ambiguous cases with real‑time AI support by providing an extra set of eyes e.g. for small, hard‑to‑spot lesions .
- Departments & Teams: Standardized EUS quality across operators, reduced variability, and faster development of junior staff.
- Hospitals & Institutions: Stronger reputation, academic prestige and leadership in pancreatic cancer care.
Together, these benefits drive more confident exams, more consistent results, and better patient outcomes — reinforcing trust and institutional excellence in pancreatic EUS. Earlier and more reliable lesion detection supports faster diagnosis, earlier treatment decisions, and ultimately improved patient pathways in a disease where time is critical.
Expert Voices
“This product will revolutionize pancreatic care going forward – saving patients’ lives and making our job as physicians easier. Medi‑Globe moved faster to market than any similar effort I’ve seen, and deserves kudos for bringing the first in the world AI augmented EUS product to market. The collaboration with our R&D team and the preeminent Scientific advisory board has been excellent from start to finish. The development of mAI Companion® should serve as a model for the future introduction of AI medical devices into interventional endoscopic practice.” — Prof. Dr. Lee Swanstroem, Interventional Endoscopist, Oregon Health and Science University, Portland Oregon USA and Director Emeritus, IHU-Strasbourg Institute of Image Guided Surgery, Strasbourg, France
“When we started the research project at the IHU in Strasbourg seven years ago, it was a dream to develop a product that would help endosonographers to do a complete pancreatic evaluation. Now that dream has become reality. I see mAI Companion as a great opportunity in medicine for more gastroenterologists to offer their patients high and consistent quality in the performance of pancreatic endoscopic ultrasound that will lead to a early diagnosis of pancreatic diseases and cancer, and I am convinced that this AI will help us doctors save patients' lives. mAI Companion will be my companion.” — Dr. Leonardo Sosa Valencia, Clinical Gastroenterologist, Digestive Echo Endoscopist, Medical Staff Manager at IHU, Strasbourg, France
"This is a major milestone for Medi-Globe. Turning mAI Companion® from concept to clinical reality required close collaboration with our partners at IHU Strasbourg and a broad international medical advisory network. Our goal is to provide clinicians with practical, workflow-integrated AI support that increases quality, confidence and consistency in pancreatic EUS." — Marc Jablonowski, Managing Director, Medi-Globe, Rohrdorf, Germany
"Delivering this programme on time and on budget reflects our disciplined approach from early prototype through MDR CE Mark, combining rigorous engineering, close clinical collaboration, and a strong focus on quality and compliance." — Dr. Markus Schönberger, President, iGlobe Scientific, Strasbourg, France
A New Era for Evidence-Based AI in Endoscopy
The launch of mAI Companion® represents a key milestone in Medi-Globe’s commitment to clinically validated, evidence-based AI solutions that can be safely integrated into routine practice, supporting earlier intervention and improved patient pathways.
With CE-marked approval, mAI Companion® is now cleared for clinical use across the EU, with initial deployments underway at leading European centres and broader rollout planned. For demonstrations or purchasing inquiries, contact Medi-Globe.
Contacts
Press OfficeMedia ContactMedi-Globe Group
press@medi-globe.deImages
Subscribe to releases from news aktuell GmbH
Subscribe to all the latest releases from news aktuell GmbH by registering your e-mail address below. You can unsubscribe at any time.
Latest releases from news aktuell GmbH
Vyoma awarded ESA data procurement contract for space-based data on small space debris24.2.2026 10:57:08 CET | Press Release
Munich, February 24th, 2026 – Vyoma, a Munich-based company providing Space Domain Awareness (SDA) capabilities, has been selected as the winner of the ESA tender on ”Data Procurement for Space-Based Statistical Data on Small Space Debris Phase 1”, funded via ESA‘s Space Safety Programme (S2P).
Grünenthal licenses exclusive Australian rights to Qutenza® to Clinect24.2.2026 08:25:47 CET | Press Release
Aachen, Germany & Victoria, Australia, 24 February 2026 – Grünenthal, a global leader in pain management and related diseases, and Clinect Pty Ltd ("Clinect"), an Australian based company focussed on supporting access to unique products, announced today that they have entered into a definitive agreement whereby Clinect will have the exclusive Australian rights to Qutenza®, a topical, non-systemic, non-opioid patch indicated for the management of peripheral neuropathic pain. Under the agreement, Clinect will be responsible for obtaining marketing authorisation for Qutenza® in Australia and, upon approval, marketing and distributing the product in Australia.
Clean energy needs a clean planet: PLAN-B NET ZERO and everwave launch joint initiative20.2.2026 13:26:45 CET | Press Release
Berlin - A strong start to 2026. PLAN-B NET ZERO enters into a partnership with the Aachen-based environmental scale-up everwave. With the guiding principle ‘1 euro = 1 kilogram of waste’, the partners recovered 10,000 kilograms of waste from rivers in Cambodia at the start of the initiative.
Grünenthal’s proprietary NaV 1.8 inhibitor enters clinical development18.2.2026 11:16:12 CET | Press Release
Aachen, Germany, 18 February 2026 – Grünenthal announced today that the first healthy volunteers have been enrolled in a Phase I trial of its voltage-gated sodium channel (NaV) 1.8 inhibitor. The orally administered investigational medicine aims to provide a non-opioid therapy option across a range of acute and chronic pain conditions. Full results of the trial are expected in the second half of 2026.
Formation of Westfalia-Automotive Group18.2.2026 08:22:54 CET | Press Release
Rheda-Wiedenbrück, Germany – the Company, one of Europe’s leading manufacturers of towing and trailering equipment as well as transport solutions, today announced the formation of Westfalia-Automotive Group (formerly Horizon Global Europe). On the 16th of February 2026, a transfer agreement (subject to fulfilment of certain conditions) has been concluded. The result of the transfer agreement is that Westfalia-Automotive Group is created, and the business is under new control and ownership. “The separation and rebranding allow for the preservation of value, but most importantly secures thousands of European manufacturing jobs and the future of the company which maintains its position as the premium brand in the towing industry. Made in Europe!” said Michael Scott, Independent Director of Westfalia-Automotive Group. As part of this transformation, you will also notice new branding being rolled out across our organisation in the next few weeks. “Our priority, as the inventor of the towbar
In our pressroom you can read all our latest releases, find our press contacts, images, documents and other relevant information about us.
Visit our pressroom